MYELOID-SPECIFIC TLR4 SIGNALING DRIVES EARLY CYTOKINE SURGE AND CD4+ T-CELL CYTOTOXICITY THAT REDUCES PANCREATIC TUMOR BURDEN IN PRECLINICAL MODELS
Utpreksha Vaish2, Suryansh Suryansh2, Dhanisha sulekha Suresh2, Maria Fernanda Noriega Salcedo2, Abhiram Beena Kannan2, Sweta Bhandari2, Tejeshwar Jain2, Srikanth Iyer2, Vikas Dudeja11. Surgery, University of Iowa Health Care, Birmingham, AL, United States.
2. The University of Alabama at Birmingham, Birmingham, AL, United States.
Introduction: Pancreatic ductal adenocarcinoma (PDAC), a lethal malignancy with a dismal 5-year overall survival is highly resistant to immunotherapy. Previously, we established that TLR4 agonism with lipopolysaccharide (LPS) mediates anti-tumor immunity against PDAC by inducing cytotoxic CD4+ T-cell responses. Here, we characterize early changes in the innate and adaptive immune events triggered by LPS administration.
Methods: KPC pancreatic cancer cells were subcutaneously implanted in syngeneic C57BL/6 (WT) mice as well as TLR4 Knockout (TLR4 KO) mice. LPS (1 mg/kg i.p.) was administered on day 10 post-implantation. Serum cytokines were analyzed by ELISA and splenic and intratumoral immune infiltration was characterized by flow cytometry. Ex-vivo CD4 cytotoxicity was assessed using a calcein release assay. The above experiment was repeated in Tlr4fl/fl LysM-Cre (TLR4-ΔMΦ) mice using WT littermates as controls and serum proteomics was performed. Additionally, tumor reducing effect of LPS was studied in the setting of specific cytokine depletion. In a preliminary cohort, BECC44, a synthetic TLR4 agonist was also evaluated for its effect on tumor growth.
Results: LPS induced a rapid spike in serum cytokines including TNF-α, IFN-γ and IL-6 within the first 6 hours, which returned to baseline by 24-48hrs. This was accompanied by expansion of Granzyme B+ CD4 T-cells in the spleen. Splenic macrophages isolated at 6 hours showed a concomitant increase in transcription of key cytokines upon LPS administration. Interestingly, TLR4-ΔMΦ mice failed to show a similar upregulation in serum cytokines after LPS injection, indicating that macrophages are the predominant source of inflammatory cytokines. Splenic CD4 T-cells from WT mice showed ex-vivo cytotoxicity upon LPS treatment, however, this effect was lost in TLR4-ΔMΦ. LPS did not show anti-tumor efficacy in TLR4-ΔMΦ, when compared to WT mice. Similarly, neutralization of TNF-α and IFN-γ abrogated the effect of LPS, implicating specific cytokines as mediators of anti-tumor response. Encouragingly, BECC44 demonstrated preliminary efficacy against PDAC with minimal systemic toxicity.
Conclusions: LPS induced TLR4 agonism can reduce tumor burden in pancreatic cancer by stimulating anti-tumor immune responses via cytotoxic CD4 T-cells and is initiated by an early increase in inflammatory cytokine secretion from myeloid cells, some of which are essential for the tumor-reducing effect of LPS and may hint at a "cytokine code" that governs anti-tumor immunity. Further investigations are required to understand the mechanisms by which these cytokines induce cytotoxic CD4 T-cells. Compounds like BECC44 are proof-of-concept that TLR4 agonists may emerge as a novel immunotherapeutic strategy against PDAC.
 Figure 1: LPS reduces pancreatic cancer burden in WT mice by increasing infiltration and cytotoxic activity of CD4+ T-cells
Figure 1: LPS reduces pancreatic cancer burden in WT mice by increasing infiltration and cytotoxic activity of CD4+ T-cells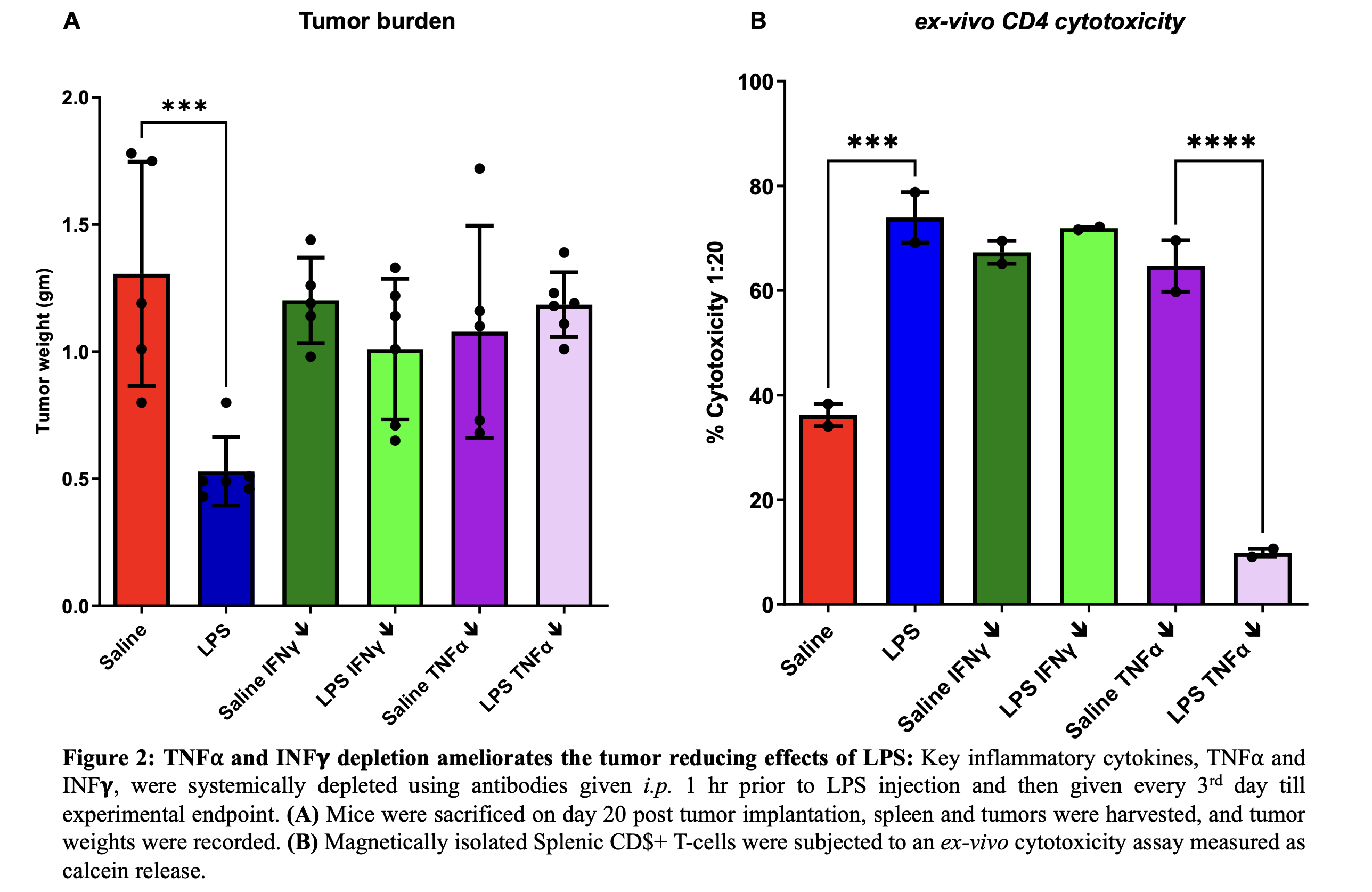 Figure 2: TNFα and INFγ depletion ameliorates the tumor reducing effects of LPS
Figure 2: TNFα and INFγ depletion ameliorates the tumor reducing effects of LPSIMAGE CAPTION: Figure 1: LPS reduces pancreatic cancer burden in WT mice by increasing infiltration and cytotoxic activity of CD4+ T-cells
Figure 2: TNFα and INFγ depletion ameliorates the tumor reducing effects of LPS
Back to 2026 Abstracts