COMPARATIVE STUDY OF ENDOLOOP-ASSISTED, FULLTHICKNESS, AND APC-ASSISTED TRANSORAL OUTLET REDUCTION IN TREATMENT FOR WEIGHT REGAIN AFTER ROUX-EN-Y GASTRIC BYPASS: A PROSPECTIVE RANDOMIZED CONTROL TRIAL PRELIMINARY RESULT
Aniwat Saleepol1, Parit Mekaroonkamol1, 2, Kittithat Tantitanawat3, Sireenada Sattawatthamrong4, Aisawan Asumpinawong5, Kasenee Tiankanon1, Phonthep Angsuwatcharakon6, 4, Pradermchai Kongkam1, 4, Suthep Udomsawaengsup5, 4, Rungsun Rerknimitr1, 41. King Chulalongkorn Memorial Hospital Department of Internal Medicine, Bangkok, Bangkok, Thailand.
2. Chulalongkorn University, Bangkok, Bangkok, Thailand.
3. Phramongkutklao Hospital, Bangkok, Bangkok, Thailand.
4. Chulalongkorn University Faculty of Medicine, Bangkok, Bangkok, Thailand.
5. King Chulalongkorn Memorial Hospital Department of Surgery, Bangkok, Bangkok, Thailand.
6. King Chulalongkorn Memorial Hospital Department of Anatomy, Bangkok, Bangkok, Thailand.
Introduction: Transoral outlet reduction (TORe) has become a standard endoscopic therapy for weight regains after Roux-en-Y gastric bypass (RYGB). Several techniques, which aim to reduce the gastrojejunal anastomotic aperture (GJA), include endoloop-assisted TORe (e-TORe), full-thickness TORe (ft-TORe), and argon plasma coagulation TORe (APC-TORe). Given the limited comparative data, this randomized controlled trial compared weight reduction efficacy among these three modalities at 12, 24, and 52 weeks.
Methods: Adults with post-RYGB weight regain > 25% from postoperative nadir, GJA > 15 mm and gastric pouch < 5 cm were randomized 1:1:1 to e-TORe, ft-TORe, or APC-TORe. One endoscopist performed all procedures. Patients were blinded to intervention and followed online weekly dietary and exercise programs. Primary outcomes were percent total weight loss (%TWL) and percent BMI reduction at 12 and 24 weeks. Secondary outcomes included %TWL and %BMI reduction at 52 weeks, percent excess weight loss, need for repeat intervention, change in GJA, and adverse events. Analyses were performed using an intention-to-treat approach.
Results: Twenty-seven patients were enrolled (e-TORe, n=10; ft-TORe, n=7; APC-TORe, n=10). Baseline characteristics, anthropometrics, cardiometabolic parameters, and GJA were comparable across groups (Table 1). At 12 weeks, e-TORe achieved the greatest %TWL (−9.05% [-10.17 to -3.49]), compared with ft-TORe (−8.47% [-10.95 to -3.49]), and APC-TORe (−3.63% [-4.98 to -1.59]) (overall p < 0.001: endoloop vs. APC, p<0.001; full-thickness vs. APC, p=0.760; endoloop vs full-thickness, p=0.933). (Table 2) At 24 weeks, e-TORe and ft-TORe maintained superior weight reduction (−4.26% and −8.80%, respectively) compared with APC-TORe (+0.85%), (overall p=0.019). By 52 weeks, differences in %TWL narrowed but remained statistically significant (p=0.049), (Table 2 and Figure1). e-TORe with repeat intervention and ft-TORe continued to demonstrate numerically greater sustained weight loss than APC group (Table 2). All techniques resulted in GJA reduction at 12-week but only repeated endoloop and full-thickness group showed sustained reduction at 52 weeks (p=0.04 and p=0.02, respectively) while APC-TORe failed to maintain GJA reduction and had the highest rate of repeat procedures (70% vs. 50% for the other groups). All techniques were safe, with mild adverse events (abdominal pain, nausea/vomiting) and no major events (bleeding, perforation, or strictures).
Conclusion: endoloop-assisted TORe demonstrated the strongest early weight loss at 12 and 24 weeks with a safety profile comparable to ft-TORe and APC-TORe. By 52 weeks, differences in %TWL decreased, yet e-TORe and ft-TORe showed more durable GJA remodeling and fewer repeat interventions than APC-TORe. Larger and longer trials are needed to confirm sustained metabolic and weight-loss benefits.
 Baseline characteristic of Endoloop-TORe, Full-thickness-TORe and APC-TORe
Baseline characteristic of Endoloop-TORe, Full-thickness-TORe and APC-TORe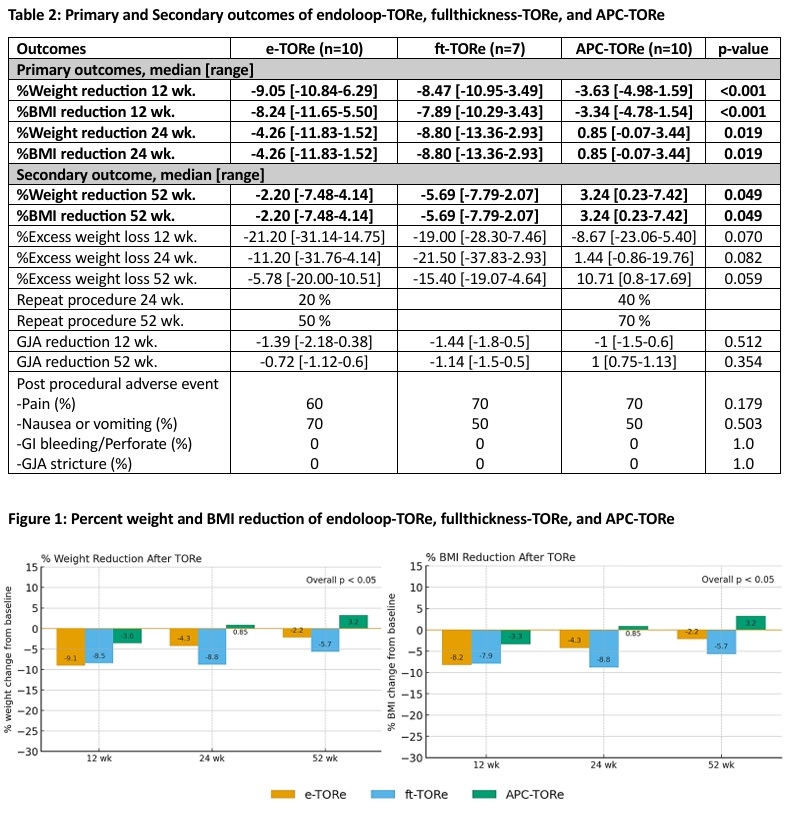 Primary and Secondary outcomes of endoloop-TORe, fullthickness-TORe, and APC-TORe
Primary and Secondary outcomes of endoloop-TORe, fullthickness-TORe, and APC-TOReIMAGE CAPTION: Baseline characteristic of Endoloop-TORe, Full-thickness-TORe and APC-TORe
Primary and Secondary outcomes of endoloop-TORe, fullthickness-TORe, and APC-TORe
Back to 2026 Abstracts