BOTULINUM TOXIN INJECTION IN THE UES FOR RETROGRADE CRICOPHARYNGEUS DYSFUNCTION (BOTUS R-CPD): FIRST RESULTS OF A TRIPLE-BLIND RANDOMIZED CONTROLLED TRIAL
Karlien Raymenants1, 2, Tim Vanuytsel2, 1, Joris Arts3, 1, Karen Van Den Houte1, Filip J Baert4, Nathalie Rommel1, Jan F. Tack1, 2, Kathelijne Delsupehe41. TARGID, Katholieke Universiteit Leuven, Leuven, Flanders, Belgium.
2. UZ Leuven, Leuven, Flanders, Belgium.
3. AZ Sint-Lucas Brugge, Bruges, Flanders, Belgium.
4. AZ Delta vzw, Roeselare, Flanders, Belgium.
Introduction
Retrograde cricopharyngeus dysfunction (R-CPD) is increasingly recognized as a distinct clinical entity characterized by a dysfunction of the belch reflex and gas-bloat symptoms. Botulinum Toxin (BT) injection in the upper esophageal sphincter (UES) has shown success in open-label case series. However, no randomized controlled trials have been performed, and data on the safety of the procedure are scarce.
Aims
To assess the efficacy and safety of BT (Botox®) injection in the UES for R-CPD in a randomized, triple-blind sham-controlled study (NCT06356025). Primary outcome was the response, defined as at least 50% improvement in ability to belch on a VAS scale ranging from 0 (no improvement) to 100 (complete resolution) at 12 weeks follow-up. Safety was an important secondary outcome. Because oropharyngeal dysphagia is a frequently reported side-effect, the Sydney Swallow Questionnaire (SSQ) was used to assess short-term safety at weeks 1 and 4.
Methods
Patients with symptoms consistent with R-CPD and typical anomalies on high resolution impedance manometry (HRiM) with belch provocation test were randomized to receive either BT 75 IU in 2mL saline or placebo (2mL saline) injected in the UES via rigid esophagoscopy during a short anesthesia. Symptoms were recorded in a newly designed questionnaire at week 1, 4, 12, 20 and 48 after the treatment, as well as safety through SSQ and recording of adverse events. HRiM was repeated 20 weeks into follow-up. If no response or early loss of response after 20 weeks, a second, open-label BT injection was proposed. The primary outcome was compared using Fisher's exact test, the difference in SSQ was compared between groups using Wilcoxon rank sum test.
Results
Fifty patients were included, and were randomized to receive either BT or placebo. Baseline characteristics were similar between groups: BT: n=25, 28% males, mean age 29 (SD 9) and BMI 23.9 (3.6) vs. Placebo: n=25, 28% males, mean age 27 (6) and BMI 22.4 (4).
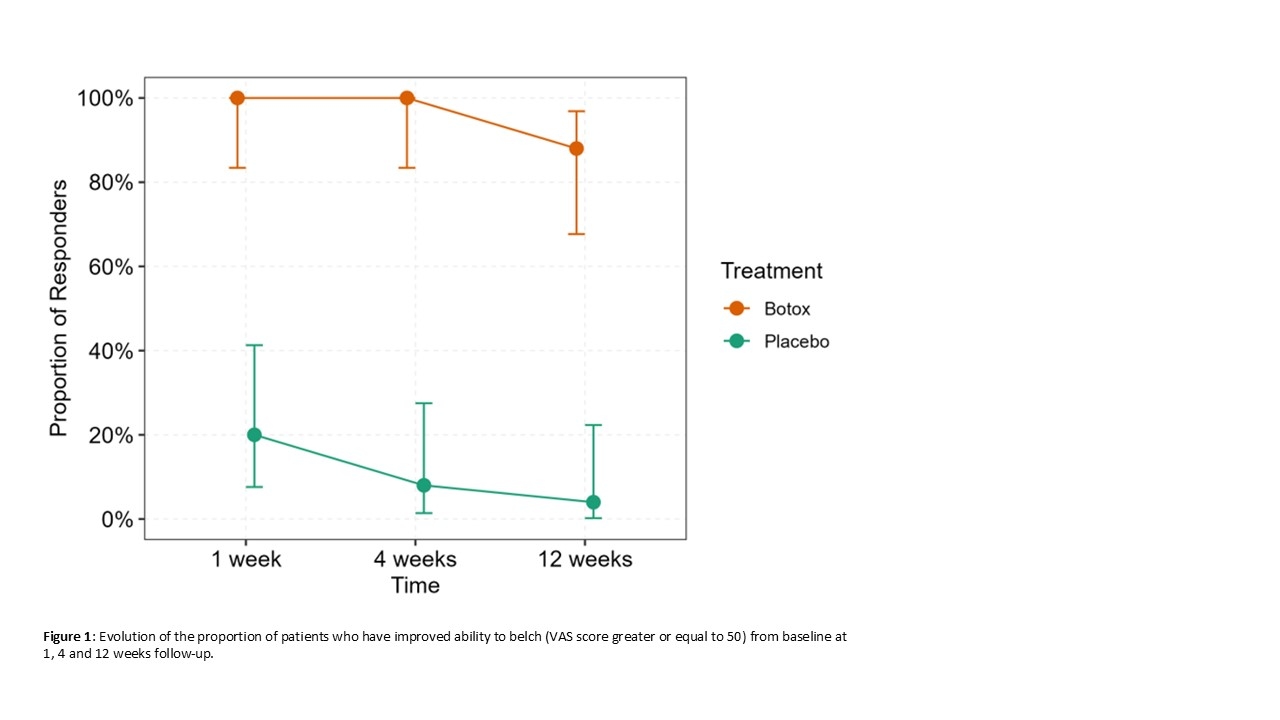
Patients who received BT had a response rate of 100% after 1 and 4 weeks, and 88% after 12 weeks. Patients in the placebo arm had 20% response at week 1, 8% at week 4, and 4% at week 12. The difference in response at 12 weeks was 84% (95% CI 62-100%, p<0.001, figure 1) (primary outcome).
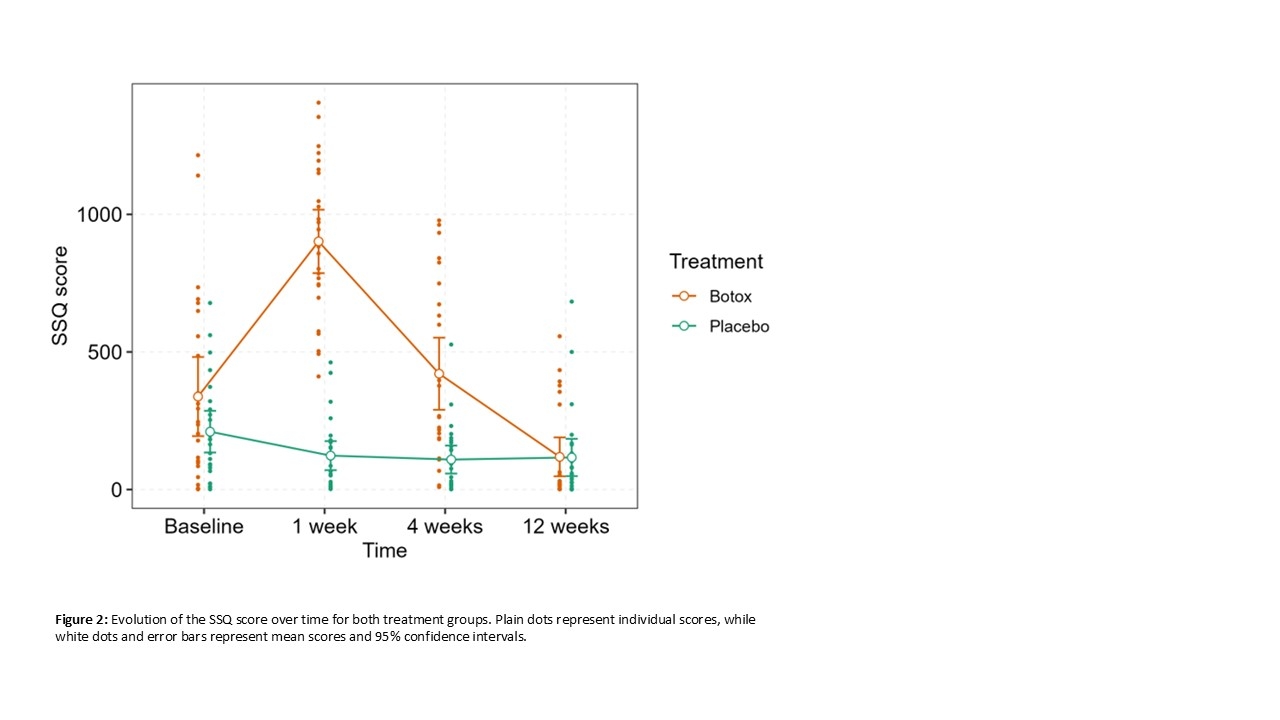
Baseline mean SSQ scores were similar between groups (338 (348) and 210 (183), p=0.4). One and 4 weeks after treatment, SSQ scores were significantly different between treatment arms, with higher SSQ scores in the group that received BT (figure 2).
Conclusion
In this randomized triple-blind controlled study, there is a clear and superior effect of BT injection in the UES over placebo on the ability to belch, along with temporary side-effects of oropharyngeal dysphagia. A longer follow-up will teach us the efficacy of a one-time injection beyond the normal duration of BT effectiveness.
Back to 2026 Abstracts