
|
 |
Back to 2015 Annual Meeting Program
Post-Operative Gastrointestinal (GI) Telemetry With a Wearable Acoustic Biosensor Distinguishes Ileus vs. Uneventful GI Recovery
Marc Kaneshiro*4, 7, William Kaiser3, Phillip Fleshner5, Marcia M. Russell2, Anne Lin6, Karen N. Zaghiyan5, Jonathan S. Pourmorady4, Bibiana M. Martinez1, Anish Patel4, Amy P. Nguyen7, Digvijay Singh3, Vince Zegarski3, Brennan Spiegel1, 2
1Health Services Research, Cedars-Sinai Center for Outcomes Research And Education (CS-CORE), Los Angeles, CA; 2Medicine, West Los Angeles VA Medical Center, Los Angeles, CA; 3Henry Samueli School of Engineering and Applied Science at UCLA, Los Angeles, CA; 4Medicine, Cedars-Sinai Medical Center, Los Angeles, CA; 5Surgery, Cedars-Sinai Medical Center, Los Angeles, CA; 6Surgery, David Geffen School of Medicine at UCLA, Los Angeles, CA; 7Medicine, David Geffen School of Medicine at UCLA, Los Angeles, CA
Background: Postoperative ileus (POI) can worsen outcomes, increase cost, and prolong hospitalization. An objective marker could help identify POI patients who should not follow standardized enhanced recovery pathways. We developed a disposable, non-invasive wearable biosensor that adheres to the external abdominal wall and monitors intra-abdominal acoustic events. Specialized software identifies stereotyped motility sounds while gating-out extraneous noise; the result is an "intestinal rate" (IR) of motility events per minute. Previous research found the sensor distinguishes POI from non-POI subjects in cross-sectional analysis (J Gastrointest Surg 2014;18:1795). The current study performed longitudinal monitoring of GI motility, akin to "GI telemetry," to examine differences in post-op motility between POI and non-POI subjects.
Methods: We studied a small, disposable sensor with a high-fidelity microphone that adheres externally to the abdominal wall (Figure 1). The device connects to a bedside computer that uses an acoustic signal processing system to measure and display motility events. We recruited subjects to wear the device both 30 minutes before and continuously after colorectal surgery. We calculated mean IR every hour until discharge and separated patients into those with vs. without POI during their post-op course. We defined POI as vomiting, need for nasogastric decompression, or imaging consistent with ileus. We used t-tests to compare: 1) mean IR in POI vs. non-POI groups across all post-op days (PODs); 2) relative change in IR from POD#1 to POD#2; and 3) across all subjects, mean IR on days with evidence of POI vs. no POI.
Results: Continuous GI telemetry was performed on 12 subjects (4 POI; 8 non-POI; age=54+13; 50%M). Figure 2 shows a sample tracing in a POI subject with persistently low IR. Mean IR was 45% lower in POI vs. non-POI groups (1.6 vs. 2.9 events/min; p<0.001). Across all subjects, IR was lower in days with POI vs. without POI (2.1 vs. 3.5 events/min; p<0.001). The first 24 hours after surgery revealed high variability in motility among subjects and no difference in IR between groups. However, in POI subjects, IR dropped by 48% between POD#1 and POD#2, whereas non-POI subjects had a 38% increase in IR during the same period (p<0.006). There were no differences in immediate pre-op IR between groups.
Conclusion: Non-invasive, abdominal acoustic monitoring with "GI telemetry" distinguishes POI from non-POI subjects. Immediate pre- and post-op motility is difficult to interpret and may not predict POI, but evidence of decreasing IR between POD#1 and #2 indicates evolving POI and should raise caution with advancing feeds. Future research will evaluate variations in post-op IR in a larger sample and determine whether "GI telemetry" can assist with safe and effective post-op decisions about feeding and timing of discharge.

Figure 1. Acoustic biosensor is size of a quarter and disposable.
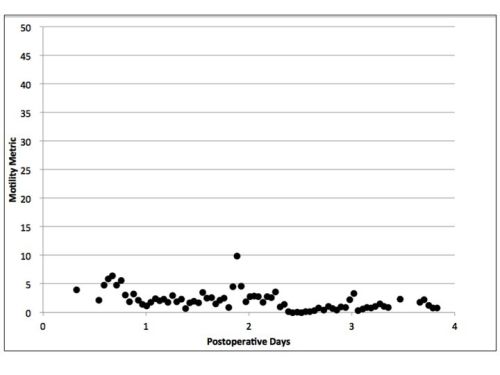
Figure 2. Example of intestinal rate (IR) measurements in POI patient.
Back to 2015 Annual Meeting Program
|



